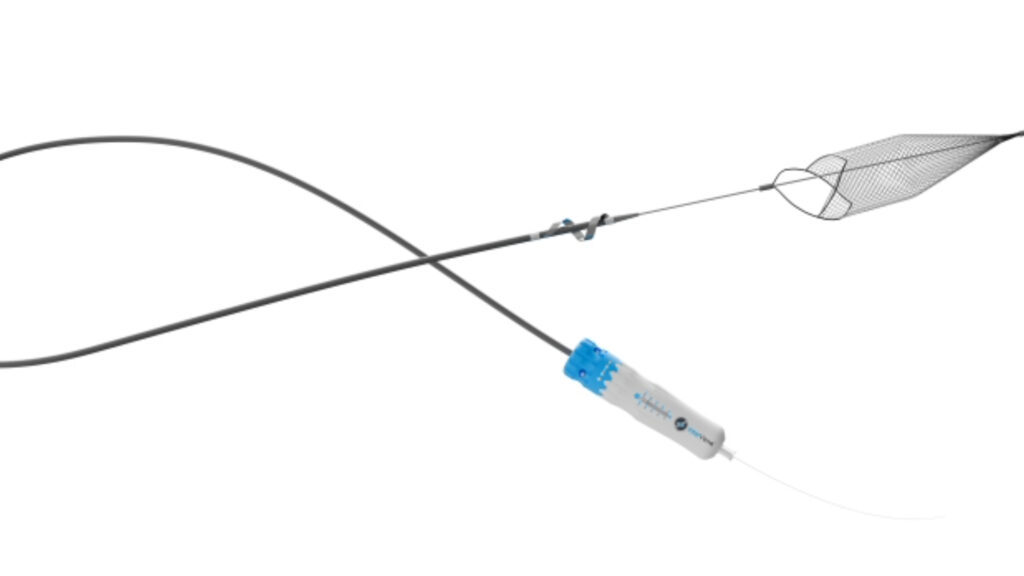
Post-thrombotic syndrome affects an estimated 20 to 50% of patients after proximal deep vein thrombosis, with severe cases in 5 to 10% of patients. Venous in-stent restenosis—a narrowing of veins where stents have been placed—poses treatment challenges for patients with deep venous conditions. InterVene, Inc., a privately held medical device company based in Redwood City, California, announced today it received 510(k) clearance from the FDA for the Recana Thrombectomy Catheter System, which the company describes as the first fully integrated platform designed for treating in-stent and native vessel obstructions.
Key Points
- The Recana system features a debulking catheter, introducer and collection sheaths, and nitinol collection baskets designed for single-session treatment of venous obstructions. The stainless-steel helical coring element has a sharpened beveled edge intended to clear venous obstructions and occlusions, with a spiral nose cone to assist with crossing.
- The system aims to reduce the need for additional devices during procedures by integrating complementary components into one platform. Nitinol collection baskets deploy from the lower extremities to capture thrombotic material.
- InterVene describes the device as addressing a critical need for treating venous in-stent restenosis, which the company characterizes as a common outcome following deep venous stent placement, particularly in patients with thrombotic pathology.
- The company is backed by RiverVest Venture Partners and Treo Ventures. Previous announcements referenced in the press release include a $13 million Series A financing round and the first patient treatment with the system.
The system is intended for physicians treating patients with venous obstructions and occlusions, particularly those with complications following stent placement.
The Data
- The announcement confirms FDA 510(k) clearance for treating venous in-stent restenosis and native vessel obstructions.
- Technical specifications mention stainless-steel construction for the coring element and nitinol material for collection baskets, though dimensional specifications were not included.
- Past research has indicated that approximately 25% of iliac venous stents require reinterventions to address in-stent restenosis or compression. A 2024 study found that venous in-stent restenosis typically occurs within one year of initial stent placement, with an incidence of 14.2% in patients undergoing iliofemoral vein stenting.
- The venous thrombectomy device market includes more than 60 FDA-approved peripheral and venous mechanical thrombectomy systems. Established competitors include Inari Medical’s ClotTriever system for deep vein thrombosis, Penumbra’s Lightning Flash system, and Boston Scientific’s AngioJet platform.
Industry Context
Venous in-stent restenosis and residual native vessel obstructions can lead to long-lasting, debilitating complications for many patients and remain difficult to treat.
Jeff Elkins, CEO of InterVene, Inc.
Venous obstructions and occlusions present challenges for traditional recanalization methods. Dr. William Marston of the University of North Carolina noted in the company announcement that such conditions can have serious long-term consequences for patients, and based on his pre-clinical experience, he views the system as a potential alternative to existing approaches. The integrated design aims to address what the company describes as limitations in current treatment options by combining multiple functions into a single system.